In 2026, waterwork utilities are facing unprecedented challenges in safeguarding our drinking water. Between emerging contaminants, evolving solutions, and limited federal action, it’s safe to say it’s uncharted territory. Contaminants that were once considered to be low priority risks are now commanding attention, shaping operational strategies, and challenging current treatment methods.
Among these, per- and polyfluoroalkyl substances (PFAS) and manganese stand out as two of the more pressing risks to our drinking water. However, they’re not the only contaminants that require our undivided attention. Tackling these contaminants requires understanding their unique properties, the risks they pose, and whether current treatment strategies are truly effective.
PFAS: The “Forever Chemicals”
PFAS are synthetic chemicals known for their strong carbon-fluorine bonds, making them heat, water, and oil resistant. While these qualities have made them popular in manufacturing, the characteristic that defines them best is persistence. Often referred to as “forever chemicals,” PFAS are not easy to break down and can accumulate in water sources and the human body over time. Despite being used for more than 80 years, PFAS are still classified as “emerging contaminants,”a term that feels misleading given their long history and lack of federal regulations on allowable limits.
In 2024, the Environmental Protection Agency (EPA) led the charge on critical treatment methods and finalized the first national drinking water standards for six PFAS compounds under the Safe Drinking Water Act. While the new regulations significantly lowered the allowable concentrations for PFAS in our drinking water — and ultimately reduced exposure and deaths — it required many utilities to implement new processes.
For utilities, the challenge is not simply compliance. PFAS exist in complex chemical cocktails, vary by water source, and require specialized treatment processes. Effective treatment relies on a combination of advanced technologies, such as activated carbon filtration, ion exchange or novel media, as well as vigorous monitoring and reporting systems and extensive regulatory frameworks to get the job done.
When treating PFAS, our team evaluates water chemistry, projected regulatory thresholds, and long-term disposal considerations before recommending any solutions. Pilot testing plays a central role, allowing systems to be optimized under real-world conditions prior to any full-scale implementation. More information on how our PFAS treatment strategies have helped our local communities can be found in the following projects:
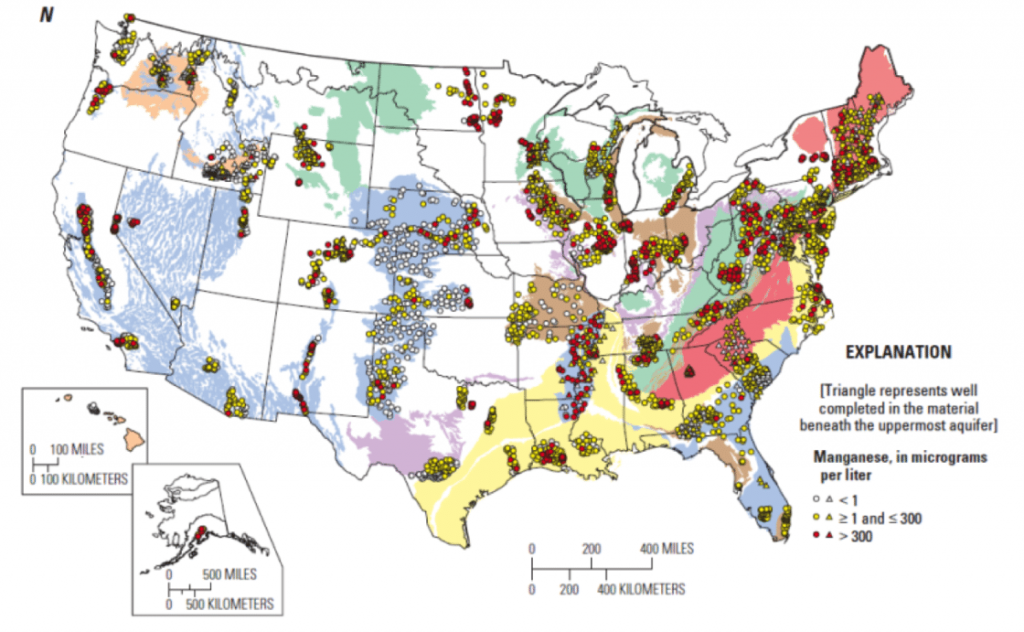
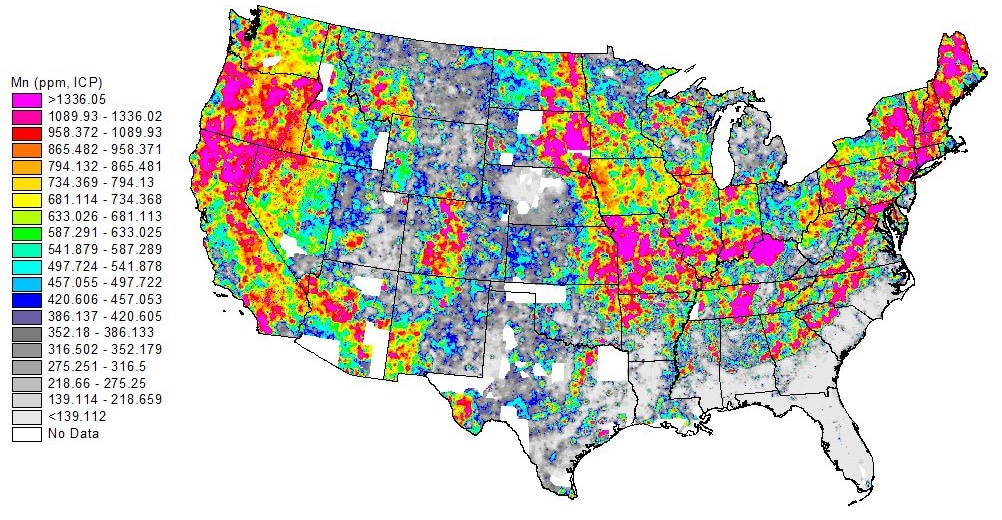
Manganese: From Aesthetic Issue to Health Concern
Unlike PFAS, manganese is not man-made but rather a naturally occurring element found in soils and rock formations. For decades, manganese was thought to be mainly an aesthetic issue, often associated with black staining, metallic taste, and buildup in pipes and delivery systems. Once considered to be safe in low concentrations, it is now under increased scrutiny as awareness of potential health impacts, particularly for infants, brings a more urgent emphasis on treatment.
 While not regulated on a federal level, the EPA does list it as a secondary contaminant and recommends a limited exposure. For manganese treatment to be effective, it must be carefully selected based on a number of factors, including concentration and water chemistry. The most common and effective treatment is a two-step process of oxidation and filtration. Oxidants such as chlorine, potassium permanganate, or ozone are used to convert soluble manganese into an insoluble form, which can then be removed through filtration. Though seemingly straightforward, it requires extensive monitoring of oxidant dosages and overall effectiveness.
While not regulated on a federal level, the EPA does list it as a secondary contaminant and recommends a limited exposure. For manganese treatment to be effective, it must be carefully selected based on a number of factors, including concentration and water chemistry. The most common and effective treatment is a two-step process of oxidation and filtration. Oxidants such as chlorine, potassium permanganate, or ozone are used to convert soluble manganese into an insoluble form, which can then be removed through filtration. Though seemingly straightforward, it requires extensive monitoring of oxidant dosages and overall effectiveness.
For manganese treatment, our team not only emphasizes extensive water quality assessments, but makes it our mission to ensure that the treatment upgrades address both compliance and safeguarding of water quality. By integrating modeling, monitoring, and operational planning, we help utilities implement solutions that are not only effective but sustainable. This level of commitment and expertise is represented in our previous work in the following communities:
Other Emerging Contaminants on the Horizon
Unfortunately, PFAS and manganese represent only a fraction of the emerging contaminants now being detected. Microplastics are now found worldwide, urging research into advanced filtration technologies. 1,4-dioxane, a groundwater solvent stabilizer, is immune to traditional adsorption (or activated carbon) methods, meaning advanced oxidation processes are required. To add, pharmaceuticals and personal care products are also being heavily scrutinized and studied, specifically when it comes to their impact by way of wastewater discharge.
Each of these contaminants carry not only unique chemical properties, but equally unique and complex treatment challenges. In 2026, utilities can no longer rely solely on decades-old treatment methods. Instead, they must adopt strategies and methods that are adaptable and supported by comprehensive data and validated by trial testing.
How T&H is Leading the Way
At Tata & Howard (T&H), we research and implement these advanced strategies for you. We stay at the forefront of this new era through extensive technical expertise and a practical, science-based, and utility-focused approach. We combine pilot testing, detailed water quality analysis, and full-scale design expertise to create treatment systems that are capable of effectively treating PFAS, manganese, and other emerging contaminants.
Beyond our projects, we contribute to industry dialogue by participating in conferences, building partnerships with regulatory and research partners, investing in data-driven decision making, and are always seeking out new innovations and technologies. This constant engagement with our industry ensures that we stay one step ahead of the curve when it comes to providing our clients with cutting edge solutions, helping them gain access to safe drinking water.
Contact us for assistance or questions about emerging contaminants or any other water quality or treatment issue. We are here to help!



 As those in the industry well know, water and wastewater treatment plants use an exorbitant amount of energy. In fact, 30-40% of total municipal energy consumption is due to water and wastewater treatment plants. In addition, energy currently accounts for 40% of drinking water systems’ operational costs and is projected to jump to 60% within the next 15 years. This excessive energy consumption places financial burden on already stressed water and wastewater utilities struggling to keep up with ever-increasing regulations and demand.
As those in the industry well know, water and wastewater treatment plants use an exorbitant amount of energy. In fact, 30-40% of total municipal energy consumption is due to water and wastewater treatment plants. In addition, energy currently accounts for 40% of drinking water systems’ operational costs and is projected to jump to 60% within the next 15 years. This excessive energy consumption places financial burden on already stressed water and wastewater utilities struggling to keep up with ever-increasing regulations and demand.




 Tata & Howard provides on-call engineering services for water, wastewater, and energy related projects for the City of Flagstaff, Arizona. Several options for replacement of the blowers were evaluated and presented to the City in a report that recommended the installation of appropriately sized turbo blowers and upgrading the controls logic to automate dissolved oxygen controls.
Tata & Howard provides on-call engineering services for water, wastewater, and energy related projects for the City of Flagstaff, Arizona. Several options for replacement of the blowers were evaluated and presented to the City in a report that recommended the installation of appropriately sized turbo blowers and upgrading the controls logic to automate dissolved oxygen controls.


 Manganese is an essential nutrient at about 2.5-5.0 mg/day, but overexposure can potentially cause serious health issues. Long term exposure to manganese can cause toxicity to the nervous system and Parkinson’s like symptoms – particularly in children, the elderly, and pregnant mothers. Young children and infants cannot break down manganese in their bodies as effectively as adults, which can cause issues in early brain development. In recent studies, children exposed to high levels of manganese experienced learning difficulties such as ADD, hyperactivity, Pervasive Development Disorder, and memory issues. Another interesting effect of overexposure to manganese is violent behavior. Studies have shown excessive manganese decreases serotonin function and reduces dopamine levels, resulting in social withdrawal, increased depression, and aggression. Studies completed in prisons have concluded manganese toxicity contributes to delinquent behavior, and autopsies of mass murderers often show toxic levels of manganese. While these studies may be concerning, manganese ingested through drinking water is processed by the liver and reduces the risks associated with other forms of manganese exposure, such as inhaling.
Manganese is an essential nutrient at about 2.5-5.0 mg/day, but overexposure can potentially cause serious health issues. Long term exposure to manganese can cause toxicity to the nervous system and Parkinson’s like symptoms – particularly in children, the elderly, and pregnant mothers. Young children and infants cannot break down manganese in their bodies as effectively as adults, which can cause issues in early brain development. In recent studies, children exposed to high levels of manganese experienced learning difficulties such as ADD, hyperactivity, Pervasive Development Disorder, and memory issues. Another interesting effect of overexposure to manganese is violent behavior. Studies have shown excessive manganese decreases serotonin function and reduces dopamine levels, resulting in social withdrawal, increased depression, and aggression. Studies completed in prisons have concluded manganese toxicity contributes to delinquent behavior, and autopsies of mass murderers often show toxic levels of manganese. While these studies may be concerning, manganese ingested through drinking water is processed by the liver and reduces the risks associated with other forms of manganese exposure, such as inhaling.
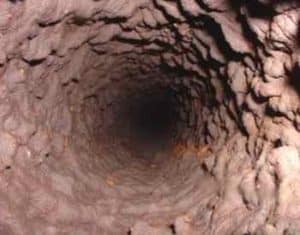 Manganese deposits can build up in pipelines, pressure tanks, water heaters, and water softeners, reducing the available quantity of the water supply and pressure in the system. Manganese accumulations can become expensive for utilities when water supply or water softening equipment must be replaced. Also, energy costs can become a burden for utilities when pumping water through constricted pipes or heating water with heating rods coated with manganese deposits. Managing safe levels of manganese in drinking water is an important step in preserving valuable assets in a distribution system. The benefits associated with treating manganese greatly outweigh the long-term repair and rehabilitation costs utilities may face with high levels of manganese. To adequately manage safe levels of manganese, proper water treatment is paramount.
Manganese deposits can build up in pipelines, pressure tanks, water heaters, and water softeners, reducing the available quantity of the water supply and pressure in the system. Manganese accumulations can become expensive for utilities when water supply or water softening equipment must be replaced. Also, energy costs can become a burden for utilities when pumping water through constricted pipes or heating water with heating rods coated with manganese deposits. Managing safe levels of manganese in drinking water is an important step in preserving valuable assets in a distribution system. The benefits associated with treating manganese greatly outweigh the long-term repair and rehabilitation costs utilities may face with high levels of manganese. To adequately manage safe levels of manganese, proper water treatment is paramount. For low concentrations of manganese, 0.3 mg/L or less, sequestering utilizing phosphate compounds is a simple, effective, and inexpensive solution. When added to water, phosphate compounds surround minerals and keep them in solution. When these compounds are put into the water system, they stabilize and disperse dissolved manganese. As a result, the manganese is not available to react with oxygen to create issues with the color, taste, or odor of drinking water. The phosphate compounds must be introduced into the water at a point where the manganese is still dissolved to maintain water clarity. This treatment process should take place before the pressure tank and as close to the well discharge point as possible. Phosphate treatment does come with a bit of risk due to the instability of most phosphate compounds at higher temperatures. If phosphate-treated water is boiled or heated, such as in a water heater, the compounds have the potential to break down and release manganese that could react with oxygen and precipitate. Also, phosphates from any source contribute to excess nutrient content in surface water.
For low concentrations of manganese, 0.3 mg/L or less, sequestering utilizing phosphate compounds is a simple, effective, and inexpensive solution. When added to water, phosphate compounds surround minerals and keep them in solution. When these compounds are put into the water system, they stabilize and disperse dissolved manganese. As a result, the manganese is not available to react with oxygen to create issues with the color, taste, or odor of drinking water. The phosphate compounds must be introduced into the water at a point where the manganese is still dissolved to maintain water clarity. This treatment process should take place before the pressure tank and as close to the well discharge point as possible. Phosphate treatment does come with a bit of risk due to the instability of most phosphate compounds at higher temperatures. If phosphate-treated water is boiled or heated, such as in a water heater, the compounds have the potential to break down and release manganese that could react with oxygen and precipitate. Also, phosphates from any source contribute to excess nutrient content in surface water.
 Ryan Neyland, P.E. Project Manager, has over 11 years of concentrated water treatment experience including all phases of planning, design, and construction services, as well as pump station rehabilitation and SCADA experience. He holds a BS in Civil Engineering from Worcester Polytechnic Institute.
Ryan Neyland, P.E. Project Manager, has over 11 years of concentrated water treatment experience including all phases of planning, design, and construction services, as well as pump station rehabilitation and SCADA experience. He holds a BS in Civil Engineering from Worcester Polytechnic Institute.